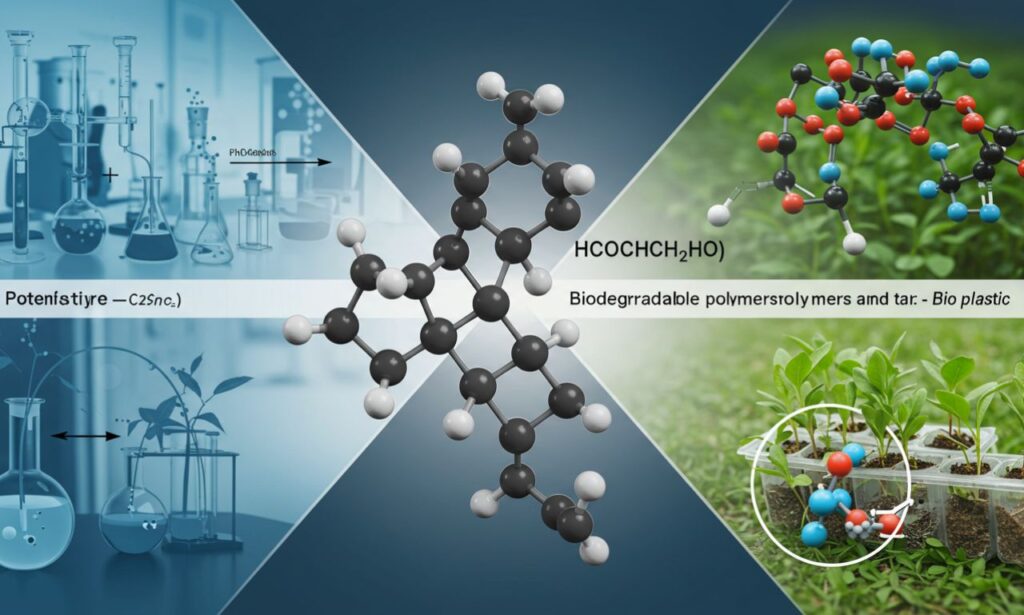
The chemical formula hcooch ch2 h2o refers to a compound related to formic acid esters and derivatives. Although it appears unusual, its structure suggests an ester linkage that can interact with water molecules. Compounds like this play an important role in organic chemistry, especially in synthesis, biochemical pathways, and industrial applications.
Chemistry often uses condensed formulas to represent molecules, and hcooch ch2 h2o is one such shorthand. To make sense of it, we break it down into components:
-
HCOOCH indicates a formate group (related to formic acid).
-
CH2 suggests a methylene group.
-
H2O represents water, which may indicate hydration or reaction conditions.
Thus, hcooch ch2 h2-o can be interpreted as an organic ester or an intermediate formed during hydrolysis or condensation reactions.
Molecular Structure of hcooch ch2 h2o
The structure of hcooch ch2 h2o can be seen as a combination of:
-
A formate ester group (HCOO–).
-
A methylene group (–CH2–).
-
A molecule of water (H2O) that is either loosely bound or present as hydration.
This makes the compound potentially reactive in hydrolysis and esterification reactions. Such structures are essential in organic synthesis since they act as intermediates for more stable products.
Bonding in hcooch ch2 h2o
The bonding within hcooch ch2 h2-o reveals interesting features:
-
C–O bonds link the formate and methylene groups.
-
Hydrogen bonding may occur between the water molecule and oxygen atoms.
-
The ester bond (C–O–C) is relatively weak and can be cleaved easily, especially in acidic or basic environments.
Understanding this bonding is vital in predicting chemical reactivity.
Formation of hcooch ch2 h2o
The compound hcooch ch2 h2-o can form in different ways depending on conditions. Some possibilities include:
-
Esterification Reaction: Formic acid reacting with methanol-like molecules, followed by hydration.
-
Hydrolysis of Esters: Breaking down formic esters in the presence of water.
-
Biochemical Pathways: Certain organisms produce similar intermediates during metabolism.
In each case, water plays a role in stabilizing or modifying the structure.
Reactions of hcooch ch2 h2o
Like many esters and hydrated intermediates, hcooch ch2 h2o participates in:
-
Hydrolysis – Water breaks down the ester bond, producing alcohol and formic acid.
-
Oxidation – Strong oxidizers convert formate into carbon dioxide.
-
Reduction – With reducing agents, it may form alcohols.
-
Condensation – Can link with other organic molecules to form polymers or complex esters.
These reactions make hcooch ch2 h2-o useful in laboratory and industrial settings.
hcooch ch2 h2o in Organic Chemistry
Organic chemists study molecules like hcooch ch2 h2o because they illustrate key concepts such as:
-
Functional groups (ester, hydroxyl).
-
Hydrolysis reactions that break down biomolecules.
-
Catalysis in acidic or enzymatic environments.
The study of such molecules improves our understanding of biochemical transformations and industrial processes.
Industrial Applications of hcooch ch2 h2-o
Industrially, compounds related to hcooch ch2 h2-o have broad applications:
-
Solvents – Esters are common in paints, coatings, and cleaning products.
-
Intermediate Chemicals – Used in producing pharmaceuticals, plastics, and polymers.
-
Preservatives – Formic derivatives can prevent microbial growth.
-
Fuel Additives – Ester-based compounds improve combustion.
Its versatility ensures relevance across industries.
hcooch ch-2 h2o in Pharmaceuticals
In pharmaceuticals, hcooch ch-2 h2o and similar esters are valuable because:
-
They act as prodrugs—inactive forms converted into active drugs in the body.
-
They serve as solubilizers for hydrophobic drugs.
-
They are intermediates in synthesizing antibiotics, painkillers, and anti-inflammatory medicines.
The ester-water bond makes them suitable for controlled release formulations.
Role of hcooch ch2 h2o in Biochemistry
In biological systems, ester-like compounds similar to hcooch ch2 h2o appear in:
-
Metabolism of fatty acids – Hydrolysis produces energy.
-
Cell signaling – Esterified molecules act as messengers.
-
Natural products – Many plant-derived molecules have similar ester structures.
This highlights its importance in life processes.
Environmental Impact of hcooch ch2 h2-o
Like many organic esters, hcooch ch-2 h2o raises environmental questions:
-
Biodegradability – Many ester-based molecules degrade naturally.
-
Water Solubility – Presence of H2O makes it likely to disperse in aquatic systems.
-
Pollution Risks – Large industrial use requires waste treatment.
Proper disposal and management ensure minimal environmental harm.
Laboratory Handling of hcooch ch2 h2-o
When dealing with hcooch ch2 h2-o in a lab:
-
Store in cool, dry places away from direct sunlight.
-
Use protective gear—gloves, goggles, and lab coats.
-
Avoid inhalation or prolonged skin contact.
-
Dispose of according to chemical safety regulations.
These safety practices are critical in research environments.
Future Potential of hcooch ch-2 h2o
Research into esters like hcooch ch2 h2o continues because they:
-
Offer green chemistry alternatives for solvents.
-
Enable biodegradable plastics.
-
Support renewable energy fuels.
-
Expand drug delivery systems.
Thus, the future of this compound lies in sustainability and innovation.
Conclusion
The chemical formula hcooch ch2 h2o may look complex, but it represents an important class of compounds—esters with hydration. These structures are central to organic chemistry, with applications in industry, pharmaceuticals, environmental science, and biochemistry.
Whether as an industrial intermediate, a pharmaceutical precursor, or a research subject, this compound plays a role in shaping modern science. Its reactivity, versatility, and potential for sustainable applications make it a fascinating topic for chemists and industry experts alike.